-
PDF
- Split View
-
Views
-
Cite
Cite
Benjamin J. Silk, Kashmira A. Date, Kelly A. Jackson, Régis Pouillot, Kristin G. Holt, Lewis M. Graves, Kanyin L. Ong, Sharon Hurd, Rebecca Meyer, Ruthanne Marcus, Beletshachew Shiferaw, Dawn M. Norton, Carlota Medus, Shelley M. Zansky, Alicia B. Cronquist, Olga L. Henao, Timothy F. Jones, Duc J. Vugia, Monica M. Farley, Barbara E. Mahon, Invasive Listeriosis in the Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009: Further Targeted Prevention Needed for Higher-Risk Groups, Clinical Infectious Diseases, Volume 54, Issue suppl_5, June 2012, Pages S396–S404, https://doi.org/10.1093/cid/cis268
Close - Share Icon Share
Abstract
Background. Listeriosis can cause severe disease, especially in fetuses, neonates, older adults, and persons with certain immunocompromising and chronic conditions. We summarize US population-based surveillance data for invasive listeriosis from 2004 through 2009.
Methods. We analyzed Foodborne Diseases Active Surveillance Network (FoodNet) data for patients with Listeria monocytogenes isolated from normally sterile sites. We describe the epidemiology of listeriosis, estimate overall and specific incidence rates, and compare pregnancy-associated and nonpregnancy-associated listeriosis by age and ethnicity.
Results. A total of 762 listeriosis cases were identified during the 6-year reporting period, including 126 pregnancy-associated cases (17%), 234 nonpregnancy-associated cases(31%) in patients aged <65 years, and 400 nonpregnancy-associated cases (53%) in patients aged ≥65 years. Eighteen percent of all cases were fatal. Meningitis was diagnosed in 44% of neonates. For 2004–2009, the overall annual incidence of listeriosis varied from 0.25 to 0.32 cases per 100 000 population. Among Hispanic women, the crude incidence of pregnancy-associated listeriosis increased from 5.09 to 12.37 cases per 100 000 for the periods of 2004–2006 and 2007–2009, respectively; among non-Hispanic women, pregnancy-associated listeriosis increased from 1.74 to 2.80 cases per 100 000 for the same periods. Incidence rates of nonpregnancy-associated listeriosis in patients aged ≥65 years were 4–5 times greater than overall rates annually.
Conclusions. Overall listeriosis incidence did not change significantly from 2004 through 2009. Further targeted prevention is needed, including food safety education and messaging (eg, avoiding Mexican-style cheese during pregnancy). Effective prevention among pregnant women, especially Hispanics, and older adults would substantially affect overall rates.
Invasive listeriosis is a severe bacterial disease caused by the consumption of food contaminated with Listeria monocytogenes. Most invasive infections (95%) are characterized by sepsis or meningoencephalitis [1]. Despite relatively low numbers of listeriosis cases in the United States (approximately 1600 annually), only nontyphoidal Salmonella and Toxoplasma gondii are estimated to be more frequent causes of death among 31 major foodborne pathogens [2].
Persons affected by invasive listeriosis are generally in established higher-risk groups [3]. Incidence rates are higher in older adults, likely due to immunosenescence of T-cell–mediated immunity and the increased prevalence of underlying diseases. Patients receiving immunosuppressive therapies and persons with impaired cell-mediated immunity or autoimmune diseases are at greater risk for invasive listeriosis [4]. In addition, several chronic diseases, including liver and end-stage renal disease, alcoholism, and diabetes mellitus [5], and other conditions (eg, achlorhydria) have been associated with an increased risk of invasive disease. Maternal infections are typically asymptomatic or mild, but pregnancy-associated listeriosis can lead to fetal loss, premature labor, and neonatal disease and death [6].
Food was first identified as the predominant route of L. monocytogenes transmission in 1981 [7]. Large outbreaks caused by ready-to-eat meats and soft cheeses, including turkey delicatessen meat, hot dogs [8, 9, 10], and Mexican-style cheese [11, 12], and the high case-fatality ratio of listeriosis, prompted a coordinated response among US regulatory and public health agencies. The Food and Drug Administration (FDA) and the US Department of Agriculture’s Food Safety and Inspection Service (USDA-FSIS) worked to improve the safety of ready-to-eat food through development of policies, guidance for the food industry, and regulations designed to prevent L. monocytogenes contamination of these foods [8]. The Centers for Disease Control and Prevention (CDC) maintains enhanced surveillance systems to facilitate prompt outbreak detection and response and to monitor incidence trends. All of these agencies also develop food safety education materials that aim to prevent listeriosis in higher-risk populations.
This report summarizes data from ongoing, active surveillance for invasive listeriosis in the United States from 2004 through 2009 [1, 13, 14]. We describe the epidemiology and incidence of pregnancy-associated and nonpregnancy-associated listeriosis by age and ethnicity.
METHODS
Surveillance for Listeriosis
The Foodborne Diseases Active Surveillance Network (FoodNet), a collaboration between CDC, 10 state health departments, USDA-FSIS, and FDA, conducts active, population-based surveillance [15]. Since 2004, FoodNet surveillance for laboratory-confirmed cases has been conducted continuously in the same geographically defined catchment area: Connecticut, Georgia, Maryland, Minnesota, New Mexico, Oregon, Tennessee, and select counties in California, Colorado, and New York [16]. FoodNet epidemiologists collect reports of all L. monocytogenes isolations in residents of the catchment area from all clinical laboratories serving this population. In 2009, the catchment area included 46.9 million persons (approximately 15% of the US population).
A case of invasive listeriosis was defined as isolation of L. monocytogenes from a normally sterile site (eg, blood or cerebrospinal fluid [CSF]) or from products of conception (eg, placental or fetal tissue). For cases in which L. monocytogenes was isolated from multiple anatomical sites, a hierarchy was applied to select the most invasive site (in descending order of invasiveness: CSF, bone or joint fluid, blood, other sterile site). Meningitis was defined by the isolation of L. monocytogenes from CSF. A pregnancy-associated case was defined as L. monocytogenes isolation from a pregnant woman, fetus, or infant aged ≤31 days. To describe the specific incidence of sporadic listeriosis in higher-risk populations, we excluded cases known to be outbreak-associated (<1% of reported cases). Available clinical isolates were forwarded by state public health laboratories to CDC for serotyping [17].
Standardized FoodNet case report forms include information on patient demographics (age, sex, ethnicity), serotyping results, and clinical characteristics (pregnancy status, whether hospitalization occurred within 7 days of specimen collection date, and vital outcome within 7 days of specimen collection date or at hospital discharge; recording of comorbid conditions was optional). State and local health departments in the FoodNet catchment area also participated in the Listeria Initiative [18], an enhanced surveillance program designed to facilitate investigation of listeriosis clusters by interviewing patients (or surrogates) to obtain detailed histories of foods consumed in the month before date of specimen collection. Listeria Initiative case reports were linked to the FoodNet dataset to improve the completeness of ethnicity, specimen source, pregnancy status, and fetal or neonatal outcome data. We calculated the frequencies of consumption of 43 selected food items that have been previously implicated as causes of listeriosis outbreaks, including ready-to-eat delicatessen meats, hot dogs, milk, and other dairy products, by risk group.
The CDC classified this work as public health surveillance, and it was therefore not subject to institutional review board review.
Statistical Analysis
We categorized cases into 1 of 3 risk groups (pregnancy-associated, nonpregnancy-associated aged <65 years, and nonpregnancy-associated aged ≥65 years) for descriptive analyses and comparison of rates by ethnicity. In this issue, Pouillot et al describe an increasing age-specific incidence of listeriosis beginning in patients aged ≥45 years [19]; however, we used 65 years as a cutoff for this analysis because incidence increases more substantially in patients aged ≥65 years. Mother–infant pairs (ie, isolation of L. monocytogenes from a clinical specimen collected from the mother and the infant) were counted as 1 case for calculating case-fatality ratios and estimating incidence. To describe seasonality, a 1-month lag in typical season groupings was used to account for the 21-day median incubation period of listeriosis (eg, summer included July, August, and September) [20]. Seasonal variations were compared with the expected mean without seasonality (ie, 25% of cases each season). Because data on comorbid conditions (immunocompromising conditions and chronic diseases) were not routinely collected and the absence of conditions could not be verified, we report the most common comorbid conditions as counts only. For our analysis, specific immunocompromising and chronic conditions included those comorbid conditions that have been described in the literature in association with invasive listeriosis risk and clinical presentation [4, 5, 21].
We used county-specific, annual population estimates from the US Census Bureau to calculate crude and adjusted incidence rates per 100 000 population. The mean annual incidence during the 3-year period from 2004 through 2006 was selected as the reference period to compare incidence changes over time. To calculate the incidence of pregnancy-associated listeriosis, we modified a published method [22] to estimate the number of Hispanic and non-Hispanic pregnant women separately using the 2005 ethnicity-specific, national fertility rates [23]. We multiplied the numbers of women of reproductive age (15–44 years) under FoodNet surveillance by nine-twelfths of their ethnicity-specific fertility rates to correct for a 9-month gestational period using annual data. We added pregnancies that ended in abortion by using published rates of induced abortion and fetal loss [23] and a one-sixth multiplier to correct for using annual data for an average 2-month incomplete gestational period. We used negative binomial regression with 95% confidence interval (CI) estimates for annual comparisons of overall incidence rates, accounting for variability in incidence among FoodNet sites [16]. We also used this approach to estimate the specific incidence of pregnancy-associated listeriosis and nonpregnancy-associated listeriosis by ethnicity and age group, respectively. Specific incidence models included terms to account for 3-year time periods (2004–2006 or 2007–2009) and variability in incidence among sites, which were categorized as lower- or higher-incidence based on the median incidence for pregnancy- and nonpregnancy-associated listeriosis; sample size precluded more specific control of incidence differences among sites.
RESULTS
From 2004 through 2009, FoodNet identified 762 sporadic cases of invasive listeriosis. Most isolates (93%) were from blood (76%) or CSF (17%) (Table 1). One-sixth of cases (n = 126) were pregnancy-associated (Table 1). Two cases in women of reproductive age were excluded from category-specific analyses because pregnancy status was unknown. Of 634 non–pregnancy-associated cases, 63% occurred in patients aged ≥65 years. Of 610 isolates available for serotyping, 577 were 4b (48%), 1/2a (29%), or 1/2b (18%). Among 533 cases with one of these serotypes and ethnicity data available, serotype 4b was significantly more common in Hispanic compared with non-Hispanic patients (55% versus 36%, respectively; P < .001). No differences in case-fatality ratio by serotype were noted (data not shown).
Demographic and Clinical Characteristics of Cases of Invasive Listeriosis, by Risk Group, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009
| Cases, No. (%) | ||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases | |||
| Characteristic | Patients Aged <65 y | Patients Aged ≥65 y | All Cases | |
| Female sex | 126 (100) | 95 (41) | 216 (54) | 401 (53) |
| Hispanic ethnicityb | 50 (42) | 26 (12) | 18 (5) | 94 (13) |
| Specimen source | ||||
| Blood | 69 (55) | 164 (70) | 348 (87) | 581 (76) |
| CSF | 31 (25) | 59 (25) | 41 (10) | 131 (17) |
| Otherc | 26 (21) | 11 (5) | 11 (3) | 48 (6) |
| Isolate serotyped | ||||
| 1/2a | 23 (18) | 52 (22) | 99 (25) | 174 (23) |
| 1/2b | 18 (14) | 32 (14) | 59 (15) | 109 (14) |
| 4b | 58 (46) | 87 (37) | 149 (37) | 294 (39) |
| Other | 4 (3) | 6 (3) | 21 (5) | 31 (4) |
| Untypable | 0 | 2 (1) | 0 | 2 (<1) |
| Unknown | 23 (18) | 55 (24) | 72 (18) | 150 (20) |
| Hospitalization | 122 (97) | 215 (92) | 378 (95) | 715 (94) |
| Death or fetal loss | 30 (29)e | 31 (13) | 79 (20) | 140 (18) |
| Total | 126 (17) | 234 (31) | 400 (53) | 760 |
| Cases, No. (%) | ||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases | |||
| Characteristic | Patients Aged <65 y | Patients Aged ≥65 y | All Cases | |
| Female sex | 126 (100) | 95 (41) | 216 (54) | 401 (53) |
| Hispanic ethnicityb | 50 (42) | 26 (12) | 18 (5) | 94 (13) |
| Specimen source | ||||
| Blood | 69 (55) | 164 (70) | 348 (87) | 581 (76) |
| CSF | 31 (25) | 59 (25) | 41 (10) | 131 (17) |
| Otherc | 26 (21) | 11 (5) | 11 (3) | 48 (6) |
| Isolate serotyped | ||||
| 1/2a | 23 (18) | 52 (22) | 99 (25) | 174 (23) |
| 1/2b | 18 (14) | 32 (14) | 59 (15) | 109 (14) |
| 4b | 58 (46) | 87 (37) | 149 (37) | 294 (39) |
| Other | 4 (3) | 6 (3) | 21 (5) | 31 (4) |
| Untypable | 0 | 2 (1) | 0 | 2 (<1) |
| Unknown | 23 (18) | 55 (24) | 72 (18) | 150 (20) |
| Hospitalization | 122 (97) | 215 (92) | 378 (95) | 715 (94) |
| Death or fetal loss | 30 (29)e | 31 (13) | 79 (20) | 140 (18) |
| Total | 126 (17) | 234 (31) | 400 (53) | 760 |
Abbreviation: CSF, cerebrospinal fluid.
Pregnancy-associated cases include those in pregnant women, fetuses, and infants aged ≤31 days. Pregnancy status was unknown for 2 women of reproductive age (ie, 2 cases excluded).
Among 703 patients (92%) with available ethnicity data.
Isolates from placenta and placental fluid (n = 20 isolates), amniotic fluid (n = 5), and chorionic membrane (n = 1) in pregnancy-associated cases and joint fluid (n = 10), peritoneal fluid (n = 6), pleural fluid (n = 3), liver (n = 2), and brain abscess (n = 1) in nonpregnancy-associated cases.
Thirty-one isolates of other Listeria monocytogenes serotypes (4%) not tabulated.
Among 102 unique, pregnancy-associated cases with fetal or neonatal outcome data available (11 mother-infant pairs counted as single cases). No deaths occurred among pregnant women.
Demographic and Clinical Characteristics of Cases of Invasive Listeriosis, by Risk Group, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009
| Cases, No. (%) | ||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases | |||
| Characteristic | Patients Aged <65 y | Patients Aged ≥65 y | All Cases | |
| Female sex | 126 (100) | 95 (41) | 216 (54) | 401 (53) |
| Hispanic ethnicityb | 50 (42) | 26 (12) | 18 (5) | 94 (13) |
| Specimen source | ||||
| Blood | 69 (55) | 164 (70) | 348 (87) | 581 (76) |
| CSF | 31 (25) | 59 (25) | 41 (10) | 131 (17) |
| Otherc | 26 (21) | 11 (5) | 11 (3) | 48 (6) |
| Isolate serotyped | ||||
| 1/2a | 23 (18) | 52 (22) | 99 (25) | 174 (23) |
| 1/2b | 18 (14) | 32 (14) | 59 (15) | 109 (14) |
| 4b | 58 (46) | 87 (37) | 149 (37) | 294 (39) |
| Other | 4 (3) | 6 (3) | 21 (5) | 31 (4) |
| Untypable | 0 | 2 (1) | 0 | 2 (<1) |
| Unknown | 23 (18) | 55 (24) | 72 (18) | 150 (20) |
| Hospitalization | 122 (97) | 215 (92) | 378 (95) | 715 (94) |
| Death or fetal loss | 30 (29)e | 31 (13) | 79 (20) | 140 (18) |
| Total | 126 (17) | 234 (31) | 400 (53) | 760 |
| Cases, No. (%) | ||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases | |||
| Characteristic | Patients Aged <65 y | Patients Aged ≥65 y | All Cases | |
| Female sex | 126 (100) | 95 (41) | 216 (54) | 401 (53) |
| Hispanic ethnicityb | 50 (42) | 26 (12) | 18 (5) | 94 (13) |
| Specimen source | ||||
| Blood | 69 (55) | 164 (70) | 348 (87) | 581 (76) |
| CSF | 31 (25) | 59 (25) | 41 (10) | 131 (17) |
| Otherc | 26 (21) | 11 (5) | 11 (3) | 48 (6) |
| Isolate serotyped | ||||
| 1/2a | 23 (18) | 52 (22) | 99 (25) | 174 (23) |
| 1/2b | 18 (14) | 32 (14) | 59 (15) | 109 (14) |
| 4b | 58 (46) | 87 (37) | 149 (37) | 294 (39) |
| Other | 4 (3) | 6 (3) | 21 (5) | 31 (4) |
| Untypable | 0 | 2 (1) | 0 | 2 (<1) |
| Unknown | 23 (18) | 55 (24) | 72 (18) | 150 (20) |
| Hospitalization | 122 (97) | 215 (92) | 378 (95) | 715 (94) |
| Death or fetal loss | 30 (29)e | 31 (13) | 79 (20) | 140 (18) |
| Total | 126 (17) | 234 (31) | 400 (53) | 760 |
Abbreviation: CSF, cerebrospinal fluid.
Pregnancy-associated cases include those in pregnant women, fetuses, and infants aged ≤31 days. Pregnancy status was unknown for 2 women of reproductive age (ie, 2 cases excluded).
Among 703 patients (92%) with available ethnicity data.
Isolates from placenta and placental fluid (n = 20 isolates), amniotic fluid (n = 5), and chorionic membrane (n = 1) in pregnancy-associated cases and joint fluid (n = 10), peritoneal fluid (n = 6), pleural fluid (n = 3), liver (n = 2), and brain abscess (n = 1) in nonpregnancy-associated cases.
Thirty-one isolates of other Listeria monocytogenes serotypes (4%) not tabulated.
Among 102 unique, pregnancy-associated cases with fetal or neonatal outcome data available (11 mother-infant pairs counted as single cases). No deaths occurred among pregnant women.
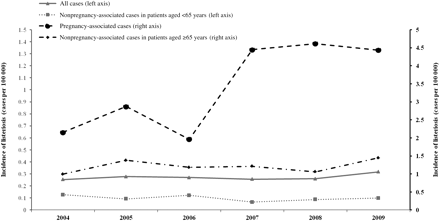
The overall annual incidence of listeriosis remained relatively constant from 2004 through 2008 (range, 0.25–0.28 cases per 100 000 population). The incidence was marginally higher in 2009 (0.32 cases per 100 000; 95% CI, .27–.37) (Figure 1), representing a 16% increase compared with 2004–2006 (0.28 per 100 000 population). Relative to the mean incidence rate from 2004 through 2006, however, modeled incidence rates did not change significantly in 2007 (95% CI, −24% to 16%), 2008 (95% CI, −20% to 22%), or 2009 (95% CI, −5% to 41%). Mean incidence rates varied from 0.12 (Minnesota) to 0.50 (Connecticut) cases of listeriosis per 100 000 population (Table 2).
Annual Frequency and Crude Incidence of Invasive Listeriosis by Risk Group and Ethnicity, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009
| Cases, No. (Crude Incidence per 100 000 Persons) | ||||||||||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases in Patients <65 y Oldb | Nonpregnancy-Associated Cases in Patients ≥65 y Oldb | All Cases | |||||||||
| Year | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc |
| 2004 | 4 (5.25) | 8 (1.67) | 12 (2.16) | 3 (0.08) | 42 (0.12) | 49 (0.13) | 2 (1.04) | 46 (0.92) | 52 (1.00) | 9 (0.22) | 96 (0.24) | 113 (0.25) |
| 2005 | 4 (5.09) | 11 (2.30) | 16 (2.87) | 6 (0.15) | 26 (0.07) | 36 (0.09) | 0 | 66 (1.30) | 73 (1.38) | 11 (0.26) | 104 (0.26) | 125 (0.28) |
| 2006 | 4 (4.92) | 6 (1.25) | 11 (1.96) | 6 (0.15) | 37 (0.10) | 48 (0.12) | 0 | 58 (1.12) | 64 (1.18) | 10 (0.23) | 101 (0.25) | 123 (0.27) |
| 2007 | 10 (11.92) | 12 (2.51) | 25 (4.45) | 3 (0.07) | 19 (0.05) | 26 (0.07) | 5 (2.22) | 55 (1.04) | 67 (1.21) | 18 (0.39) | 86 (0.21) | 118 (0.26) |
| 2008 | 10 (11.60) | 16 (3.35) | 26 (4.61) | 3 (0.07) | 28 (0.08) | 35 (0.09) | 8 (3.36) | 50 (0.92) | 60 (1.06) | 21 (0.44) | 94 (0.23) | 121 (0.26) |
| 2009 | 12 (13.58 | 12 (2.53) | 25 (4.44) | 5 (0.11) | 33 (0.09) | 40 (0.10) | 3 (1.20) | 75 (1.35) | 84 (1.45) | 20 (0.40) | 120 (0.29) | 149 (0.32) |
| Total | 44 (8.90) | 65 (2.27) | 115 (3.42) | 26 (0.10) | 185 (0.09) | 234 (0.10) | 18 (1.36) | 350 (1.11) | 400 (1.21) | 89 (0.33) | 601 (0.24) | 749 (0.27) |
| State variability | ||||||||||||
| Total casesd | 0–17 | 0–22 | 1–40 | 0–9 | 2–28 | 6–37 | 0–6 | 15–57 | 16–64 | 0–24 | 9–103 | 23–134 |
| Incidenced | 0–32.10 | 0–3.44 | 0.27–5.61 | 0–0.40 | 0.03–0.16 | 0.05–0.18 | 0–3.65 | 0.58–2.09 | 0.60–2.22 | 0–0.61 | 0.12–0.46 | 0.12–0.50 |
| Cases, No. (Crude Incidence per 100 000 Persons) | ||||||||||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases in Patients <65 y Oldb | Nonpregnancy-Associated Cases in Patients ≥65 y Oldb | All Cases | |||||||||
| Year | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc |
| 2004 | 4 (5.25) | 8 (1.67) | 12 (2.16) | 3 (0.08) | 42 (0.12) | 49 (0.13) | 2 (1.04) | 46 (0.92) | 52 (1.00) | 9 (0.22) | 96 (0.24) | 113 (0.25) |
| 2005 | 4 (5.09) | 11 (2.30) | 16 (2.87) | 6 (0.15) | 26 (0.07) | 36 (0.09) | 0 | 66 (1.30) | 73 (1.38) | 11 (0.26) | 104 (0.26) | 125 (0.28) |
| 2006 | 4 (4.92) | 6 (1.25) | 11 (1.96) | 6 (0.15) | 37 (0.10) | 48 (0.12) | 0 | 58 (1.12) | 64 (1.18) | 10 (0.23) | 101 (0.25) | 123 (0.27) |
| 2007 | 10 (11.92) | 12 (2.51) | 25 (4.45) | 3 (0.07) | 19 (0.05) | 26 (0.07) | 5 (2.22) | 55 (1.04) | 67 (1.21) | 18 (0.39) | 86 (0.21) | 118 (0.26) |
| 2008 | 10 (11.60) | 16 (3.35) | 26 (4.61) | 3 (0.07) | 28 (0.08) | 35 (0.09) | 8 (3.36) | 50 (0.92) | 60 (1.06) | 21 (0.44) | 94 (0.23) | 121 (0.26) |
| 2009 | 12 (13.58 | 12 (2.53) | 25 (4.44) | 5 (0.11) | 33 (0.09) | 40 (0.10) | 3 (1.20) | 75 (1.35) | 84 (1.45) | 20 (0.40) | 120 (0.29) | 149 (0.32) |
| Total | 44 (8.90) | 65 (2.27) | 115 (3.42) | 26 (0.10) | 185 (0.09) | 234 (0.10) | 18 (1.36) | 350 (1.11) | 400 (1.21) | 89 (0.33) | 601 (0.24) | 749 (0.27) |
| State variability | ||||||||||||
| Total casesd | 0–17 | 0–22 | 1–40 | 0–9 | 2–28 | 6–37 | 0–6 | 15–57 | 16–64 | 0–24 | 9–103 | 23–134 |
| Incidenced | 0–32.10 | 0–3.44 | 0.27–5.61 | 0–0.40 | 0.03–0.16 | 0.05–0.18 | 0–3.65 | 0.58–2.09 | 0.60–2.22 | 0–0.61 | 0.12–0.46 | 0.12–0.50 |
Pregnancy-associated cases include cases in pregnant women, fetuses, and infants aged ≤31 days. Eleven mother–baby pairs counted as single cases.
Eight nonpregnancy-associated cases known to be associated with outbreaks in New York in 2004 (n = 1), 2005 (n = 2), or 2006 (n = 1) or Oregon in 2006 (n = 4) were excluded.
Data on all patients include cases with missing ethnicity data (n = 61).
Range in the total case counts and average annual incidence for 2004–2009 among 10 FoodNet states (California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee).
Annual Frequency and Crude Incidence of Invasive Listeriosis by Risk Group and Ethnicity, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009
| Cases, No. (Crude Incidence per 100 000 Persons) | ||||||||||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases in Patients <65 y Oldb | Nonpregnancy-Associated Cases in Patients ≥65 y Oldb | All Cases | |||||||||
| Year | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc |
| 2004 | 4 (5.25) | 8 (1.67) | 12 (2.16) | 3 (0.08) | 42 (0.12) | 49 (0.13) | 2 (1.04) | 46 (0.92) | 52 (1.00) | 9 (0.22) | 96 (0.24) | 113 (0.25) |
| 2005 | 4 (5.09) | 11 (2.30) | 16 (2.87) | 6 (0.15) | 26 (0.07) | 36 (0.09) | 0 | 66 (1.30) | 73 (1.38) | 11 (0.26) | 104 (0.26) | 125 (0.28) |
| 2006 | 4 (4.92) | 6 (1.25) | 11 (1.96) | 6 (0.15) | 37 (0.10) | 48 (0.12) | 0 | 58 (1.12) | 64 (1.18) | 10 (0.23) | 101 (0.25) | 123 (0.27) |
| 2007 | 10 (11.92) | 12 (2.51) | 25 (4.45) | 3 (0.07) | 19 (0.05) | 26 (0.07) | 5 (2.22) | 55 (1.04) | 67 (1.21) | 18 (0.39) | 86 (0.21) | 118 (0.26) |
| 2008 | 10 (11.60) | 16 (3.35) | 26 (4.61) | 3 (0.07) | 28 (0.08) | 35 (0.09) | 8 (3.36) | 50 (0.92) | 60 (1.06) | 21 (0.44) | 94 (0.23) | 121 (0.26) |
| 2009 | 12 (13.58 | 12 (2.53) | 25 (4.44) | 5 (0.11) | 33 (0.09) | 40 (0.10) | 3 (1.20) | 75 (1.35) | 84 (1.45) | 20 (0.40) | 120 (0.29) | 149 (0.32) |
| Total | 44 (8.90) | 65 (2.27) | 115 (3.42) | 26 (0.10) | 185 (0.09) | 234 (0.10) | 18 (1.36) | 350 (1.11) | 400 (1.21) | 89 (0.33) | 601 (0.24) | 749 (0.27) |
| State variability | ||||||||||||
| Total casesd | 0–17 | 0–22 | 1–40 | 0–9 | 2–28 | 6–37 | 0–6 | 15–57 | 16–64 | 0–24 | 9–103 | 23–134 |
| Incidenced | 0–32.10 | 0–3.44 | 0.27–5.61 | 0–0.40 | 0.03–0.16 | 0.05–0.18 | 0–3.65 | 0.58–2.09 | 0.60–2.22 | 0–0.61 | 0.12–0.46 | 0.12–0.50 |
| Cases, No. (Crude Incidence per 100 000 Persons) | ||||||||||||
| Pregnancy-Associated Casesa | Nonpregnancy-Associated Cases in Patients <65 y Oldb | Nonpregnancy-Associated Cases in Patients ≥65 y Oldb | All Cases | |||||||||
| Year | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc | Hispanic Patients | Non-Hispanic Patients | All Patientsc |
| 2004 | 4 (5.25) | 8 (1.67) | 12 (2.16) | 3 (0.08) | 42 (0.12) | 49 (0.13) | 2 (1.04) | 46 (0.92) | 52 (1.00) | 9 (0.22) | 96 (0.24) | 113 (0.25) |
| 2005 | 4 (5.09) | 11 (2.30) | 16 (2.87) | 6 (0.15) | 26 (0.07) | 36 (0.09) | 0 | 66 (1.30) | 73 (1.38) | 11 (0.26) | 104 (0.26) | 125 (0.28) |
| 2006 | 4 (4.92) | 6 (1.25) | 11 (1.96) | 6 (0.15) | 37 (0.10) | 48 (0.12) | 0 | 58 (1.12) | 64 (1.18) | 10 (0.23) | 101 (0.25) | 123 (0.27) |
| 2007 | 10 (11.92) | 12 (2.51) | 25 (4.45) | 3 (0.07) | 19 (0.05) | 26 (0.07) | 5 (2.22) | 55 (1.04) | 67 (1.21) | 18 (0.39) | 86 (0.21) | 118 (0.26) |
| 2008 | 10 (11.60) | 16 (3.35) | 26 (4.61) | 3 (0.07) | 28 (0.08) | 35 (0.09) | 8 (3.36) | 50 (0.92) | 60 (1.06) | 21 (0.44) | 94 (0.23) | 121 (0.26) |
| 2009 | 12 (13.58 | 12 (2.53) | 25 (4.44) | 5 (0.11) | 33 (0.09) | 40 (0.10) | 3 (1.20) | 75 (1.35) | 84 (1.45) | 20 (0.40) | 120 (0.29) | 149 (0.32) |
| Total | 44 (8.90) | 65 (2.27) | 115 (3.42) | 26 (0.10) | 185 (0.09) | 234 (0.10) | 18 (1.36) | 350 (1.11) | 400 (1.21) | 89 (0.33) | 601 (0.24) | 749 (0.27) |
| State variability | ||||||||||||
| Total casesd | 0–17 | 0–22 | 1–40 | 0–9 | 2–28 | 6–37 | 0–6 | 15–57 | 16–64 | 0–24 | 9–103 | 23–134 |
| Incidenced | 0–32.10 | 0–3.44 | 0.27–5.61 | 0–0.40 | 0.03–0.16 | 0.05–0.18 | 0–3.65 | 0.58–2.09 | 0.60–2.22 | 0–0.61 | 0.12–0.46 | 0.12–0.50 |
Pregnancy-associated cases include cases in pregnant women, fetuses, and infants aged ≤31 days. Eleven mother–baby pairs counted as single cases.
Eight nonpregnancy-associated cases known to be associated with outbreaks in New York in 2004 (n = 1), 2005 (n = 2), or 2006 (n = 1) or Oregon in 2006 (n = 4) were excluded.
Data on all patients include cases with missing ethnicity data (n = 61).
Range in the total case counts and average annual incidence for 2004–2009 among 10 FoodNet states (California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee).
Crude incidence of invasive listeriosis over time by risk group, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009. Note: The apparent temporal increases in the crude incidence of pregnancy-associated listeriosis were not statistically significant when adjusted incidence rates were modeled for Hispanic and non-Hispanic pregnant women.
Pregnancy-Associated Listeriosis
L. monocytogenes was isolated from 68 infants (54% of pregnancy-associated cases). Among neonates, listeriosis was diagnosed as early as 1 day (41%), and usually within 1 week (51%) or 2 weeks (78%) after birth; 44% had meningitis. The median age of when 30 infants with meningitis were diagnosed was 15 days (interquartile range, 13–20 days) compared with 1 day (interquartile range, 1–2 days) in 38 neonates without this diagnosis. Of 102 cases with outcome data available, 30 resulted in fetal losses or neonatal death (case-fatality ratio, 29%). No deaths among infants with meningitis or among pregnant women were reported.
Among 701 (92%) cases with ethnicity data available, a significantly larger proportion of pregnancy-associated cases occurred among Hispanic patients compared with nonpregnancy-associated cases (42% versus 8%, respectively; P < .0001). Among 701 (92%) cases with ethnicity data available, Hispanic patients composed a significantly larger proportion of pregnancy-associated cases than they did of nonpregnancy-associated cases (42% versus 8%, respectively; P = .0001). Fetal loss or neonatal death was not associated with ethnicity (P = .35) or serotype (P = .50). Four pregnant women each reported a comorbid condition (diabetes mellitus, human immunodeficiency virus [HIV] infection, malignancy, and renal failure).
Crude rates of pregnancy-associated listeriosis were 8–18 times greater than crude rates of overall listeriosis annually from 2004 through 2009 (Table 2). The highest rates of listeriosis occurred among Hispanic pregnant women during the 2007–2009 period (3-year mean incidence, 12.40 cases per 100 000 Hispanic pregnant women), which was more than double the mean rate during 2004–2006 (5.09 cases per 100 000) (Table 2). Corresponding 3-year mean rates among non-Hispanic pregnant women were also higher for 2007–2009 compared with 2004–2006 (2.80 versus 1.74 cases per 100 000 non-Hispanic pregnant women). These increases were largely attributable to an increase in cases among Hispanic pregnant women in Georgia (14 of 44 cases [32%]) and non-Hispanic pregnant women in Georgia and New York (20 of 65 cases [31%]) during 2007–2009.
The mean annual incidence rates of pregnancy-associated listeriosis varied by FoodNet site from 0.27 (Minnesota) to 5.61 (Georgia) cases per 100 000 pregnant women. Variability in the population percentages of Hispanics (range, 4%–45%) did not correlate with the incidence of pregnancy-associated listeriosis (eg, New Mexico is 45% Hispanic but had the second lowest incidence). The apparent temporal increases in the crude incidence of pregnancy-associated listeriosis (Table 2 and Figure 1) were not statistically significant when adjusted incidence rates were modeled for Hispanic and non-Hispanic pregnant women.
Nonpregnancy-Associated Listeriosis
Listeria monocytogenes was isolated from blood (n = 512) or CSF (n = 100) in 97% of nonpregnancy-associated cases. The median age of patients with bacteremia was 71.5 years, with the following age distribution: 1–17 years, 1%; 18–49 years, 11%; 50–64 years, 21%; and ≥65 years, 68%. Patients with meningitis were generally younger (median, 61 years), with the following age group distribution: 1–17 years, 6%; 18–49 years, 20%; 50–64 years, 33%; and ≥65 years, 41%. Overall, the median age was older for female than male patients with nonpregnancy-associated listeriosis (72 versus 67 years, respectively; P = .002).
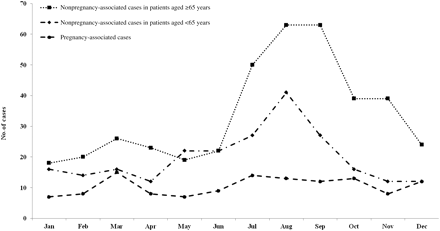
Among the nonpregnancy-associated cases, 443 (70%) had ≥1 comorbid conditions recorded, most frequently cancer or malignancy (n = 104), immunosuppressive therapy (including steroid treatment, chemotherapy, and radiation) (n = 81), diabetes mellitus (n = 63), renal failure or dialysis (n = 48), atherosclerosis (n = 46), and congestive heart failure or unspecified heart disease (n = 46). Alcohol abuse (n = 26); cirrhosis, liver failure, or hepatitis (n = 25); and HIV/AIDS (n = 16) were also recorded. Among meningitis patients with ≥1 comorbid condition, a fatal outcome was more common in patients aged ≥65 years (29%) compared with persons aged <65 years (4%) (P = .02). In contrast, for patients with ≥1 comorbid condition and bacteremia, case-fatality ratios did not differ significantly by age group (P = .5). Nonpregnancy-associated cases were more often diagnosed in July, August, or September (42%; P < .0001) (Figure 2) with relatively consistent increases among males (39%), females (45%), Hispanics (51%), and non-Hispanics (42%).
Number of invasive listeriosis cases by month and risk group, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009.
Crude rates of nonpregnancy-associated listeriosis in patients aged ≥65 years were 4–5 times greater than the crude rates of overall listeriosis annually during the 6-year reporting period (Table 2). Among the FoodNet sites, the mean annual incidence rates of nonpregnancy-associated listeriosis varied from 0.12 (Minnesota) to 0.46 (Connecticut) cases per 100 000 population, although there was relatively little variability in the population percentages of adults aged ≥65 years (range, 10%–15%).
Consumption of Selected Foods
Food consumption histories in the month preceding date of specimen collection were available for 568 patients (75%) (Table 3). The average number of 43 food items consumed was 9.0 (standard deviation, 4.5) and was similar by risk group and ethnicity. Among pregnant women, non-Hispanics were significantly more likely than Hispanics to report consuming turkey breast delicatessen meat (36% versus 16%, respectively; P = .01) and hot dogs (46% versus 22%, respectively; P = .01). Consumption of Mexican-style cheese was more frequently reported by Hispanic pregnant women (58% versus 16%, respectively; P < .0001). Among nonpregnant patients, Hispanics also were more likely than non-Hispanics to report consumption of Mexican-style cheese (31% versus 4%, respectively; P < .0001). Age group differences in the frequency of food consumption of other foods were not observed among nonpregnant patients.
Consumption of Selected Foods Among Cases of Invasive Listeriosis by Risk Group and Ethnicity, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009
| Cases, No. (%) | |||||||||
| Pregnancy-Associated Casesb | Nonpregnancy-Associated Cases | All Cases | |||||||
| Selected Fooda | Hispanic Patients | Non-Hispanic Patients | All Patients | Patients Aged <65 y | Patients Aged ≥65 y | All Patients | Hispanic Patients | Non-Hispanic Patients | All Patients |
| Deli meat/hot dogs | |||||||||
| Turkey breastc | 8 (16) | 25 (36) | 35 (28) | 66 (28) | 91 (23) | 157 (25) | 17 (18) | 160 (28) | 192 (25) |
| Chicken | 14 (28) | 12 (17) | 26 (21) | 31 (13) | 38 (10) | 69 (11) | 21 (22) | 70 (12) | 95 (12) |
| Hot dogsc | 11 (22) | 32 (46) | 45 (36) | 73 (31) | 124 (31) | 197 (31) | 20 (21) | 204 (34) | 242 (32) |
| Cheese, milk, yogurt | |||||||||
| Mexican-style cheesed | 29 (58) | 11 (16) | 41 (33) | 21 (9) | 19 (5) | 40 (6) | 43 (45) | 33 (5) | 81 (11) |
| Whole milkc | 29 (58) | 25 (36) | 56 (44) | 38 (16) | 55 (14) | 93 (15) | 37 (39) | 106 (17) | 149 (20) |
| 2% milkc | 6 (12) | 21 (30) | 29 (23) | 48 (21) | 100 (25) | 148 (23) | 16 (17) | 154 (25) | 177 (23) |
| Yogurt | 23 (46) | 21 (30) | 46 (37) | 54 (23) | 99 (25) | 153 (24) | 29 (31) | 155 (26) | 199 (26) |
| No. of food historiese | 42 (84) | 51 (73) | 96 (76) | 171 (73) | 301 (75) | 472 (74) | 71 (75) | 468 (77) | 568 (75) |
| Cases, No. (%) | |||||||||
| Pregnancy-Associated Casesb | Nonpregnancy-Associated Cases | All Cases | |||||||
| Selected Fooda | Hispanic Patients | Non-Hispanic Patients | All Patients | Patients Aged <65 y | Patients Aged ≥65 y | All Patients | Hispanic Patients | Non-Hispanic Patients | All Patients |
| Deli meat/hot dogs | |||||||||
| Turkey breastc | 8 (16) | 25 (36) | 35 (28) | 66 (28) | 91 (23) | 157 (25) | 17 (18) | 160 (28) | 192 (25) |
| Chicken | 14 (28) | 12 (17) | 26 (21) | 31 (13) | 38 (10) | 69 (11) | 21 (22) | 70 (12) | 95 (12) |
| Hot dogsc | 11 (22) | 32 (46) | 45 (36) | 73 (31) | 124 (31) | 197 (31) | 20 (21) | 204 (34) | 242 (32) |
| Cheese, milk, yogurt | |||||||||
| Mexican-style cheesed | 29 (58) | 11 (16) | 41 (33) | 21 (9) | 19 (5) | 40 (6) | 43 (45) | 33 (5) | 81 (11) |
| Whole milkc | 29 (58) | 25 (36) | 56 (44) | 38 (16) | 55 (14) | 93 (15) | 37 (39) | 106 (17) | 149 (20) |
| 2% milkc | 6 (12) | 21 (30) | 29 (23) | 48 (21) | 100 (25) | 148 (23) | 16 (17) | 154 (25) | 177 (23) |
| Yogurt | 23 (46) | 21 (30) | 46 (37) | 54 (23) | 99 (25) | 153 (24) | 29 (31) | 155 (26) | 199 (26) |
| No. of food historiese | 42 (84) | 51 (73) | 96 (76) | 171 (73) | 301 (75) | 472 (74) | 71 (75) | 468 (77) | 568 (75) |
Self-reported consumption of food in the 4 weeks before interview. Data include cases with missing ethnicity data (n = 61).
Pregnancy-associated cases include cases in pregnant women, fetuses, and infants aged ≤31 days.
Among pregnant patients, significant difference in frequency of consumption of a selected foods by ethnicity (P < .05).
Among pregnant and nonpregnant patients, significant difference in frequency of consumption of Mexican-style cheese by ethnicity (P < .0001).
Total number of food histories includes partial histories; totals are not a comparable denominator for specific frequencies owing to item nonresponse.
Consumption of Selected Foods Among Cases of Invasive Listeriosis by Risk Group and Ethnicity, Foodborne Diseases Active Surveillance Network (FoodNet), 2004–2009
| Cases, No. (%) | |||||||||
| Pregnancy-Associated Casesb | Nonpregnancy-Associated Cases | All Cases | |||||||
| Selected Fooda | Hispanic Patients | Non-Hispanic Patients | All Patients | Patients Aged <65 y | Patients Aged ≥65 y | All Patients | Hispanic Patients | Non-Hispanic Patients | All Patients |
| Deli meat/hot dogs | |||||||||
| Turkey breastc | 8 (16) | 25 (36) | 35 (28) | 66 (28) | 91 (23) | 157 (25) | 17 (18) | 160 (28) | 192 (25) |
| Chicken | 14 (28) | 12 (17) | 26 (21) | 31 (13) | 38 (10) | 69 (11) | 21 (22) | 70 (12) | 95 (12) |
| Hot dogsc | 11 (22) | 32 (46) | 45 (36) | 73 (31) | 124 (31) | 197 (31) | 20 (21) | 204 (34) | 242 (32) |
| Cheese, milk, yogurt | |||||||||
| Mexican-style cheesed | 29 (58) | 11 (16) | 41 (33) | 21 (9) | 19 (5) | 40 (6) | 43 (45) | 33 (5) | 81 (11) |
| Whole milkc | 29 (58) | 25 (36) | 56 (44) | 38 (16) | 55 (14) | 93 (15) | 37 (39) | 106 (17) | 149 (20) |
| 2% milkc | 6 (12) | 21 (30) | 29 (23) | 48 (21) | 100 (25) | 148 (23) | 16 (17) | 154 (25) | 177 (23) |
| Yogurt | 23 (46) | 21 (30) | 46 (37) | 54 (23) | 99 (25) | 153 (24) | 29 (31) | 155 (26) | 199 (26) |
| No. of food historiese | 42 (84) | 51 (73) | 96 (76) | 171 (73) | 301 (75) | 472 (74) | 71 (75) | 468 (77) | 568 (75) |
| Cases, No. (%) | |||||||||
| Pregnancy-Associated Casesb | Nonpregnancy-Associated Cases | All Cases | |||||||
| Selected Fooda | Hispanic Patients | Non-Hispanic Patients | All Patients | Patients Aged <65 y | Patients Aged ≥65 y | All Patients | Hispanic Patients | Non-Hispanic Patients | All Patients |
| Deli meat/hot dogs | |||||||||
| Turkey breastc | 8 (16) | 25 (36) | 35 (28) | 66 (28) | 91 (23) | 157 (25) | 17 (18) | 160 (28) | 192 (25) |
| Chicken | 14 (28) | 12 (17) | 26 (21) | 31 (13) | 38 (10) | 69 (11) | 21 (22) | 70 (12) | 95 (12) |
| Hot dogsc | 11 (22) | 32 (46) | 45 (36) | 73 (31) | 124 (31) | 197 (31) | 20 (21) | 204 (34) | 242 (32) |
| Cheese, milk, yogurt | |||||||||
| Mexican-style cheesed | 29 (58) | 11 (16) | 41 (33) | 21 (9) | 19 (5) | 40 (6) | 43 (45) | 33 (5) | 81 (11) |
| Whole milkc | 29 (58) | 25 (36) | 56 (44) | 38 (16) | 55 (14) | 93 (15) | 37 (39) | 106 (17) | 149 (20) |
| 2% milkc | 6 (12) | 21 (30) | 29 (23) | 48 (21) | 100 (25) | 148 (23) | 16 (17) | 154 (25) | 177 (23) |
| Yogurt | 23 (46) | 21 (30) | 46 (37) | 54 (23) | 99 (25) | 153 (24) | 29 (31) | 155 (26) | 199 (26) |
| No. of food historiese | 42 (84) | 51 (73) | 96 (76) | 171 (73) | 301 (75) | 472 (74) | 71 (75) | 468 (77) | 568 (75) |
Self-reported consumption of food in the 4 weeks before interview. Data include cases with missing ethnicity data (n = 61).
Pregnancy-associated cases include cases in pregnant women, fetuses, and infants aged ≤31 days.
Among pregnant patients, significant difference in frequency of consumption of a selected foods by ethnicity (P < .05).
Among pregnant and nonpregnant patients, significant difference in frequency of consumption of Mexican-style cheese by ethnicity (P < .0001).
Total number of food histories includes partial histories; totals are not a comparable denominator for specific frequencies owing to item nonresponse.
DISCUSSION
The incidence of invasive listeriosis among pregnant women remains much higher than in the overall population, indicating that effective prevention in this group would have a substantial impact on overall rates. In addition, the incidence in pregnant women was substantially higher each year from 2007 through 2009 than in any other year since 2001 [1]. The average incidence rate of pregnancy-associated listeriosis from 2004 through 2009 was 4.5 cases per 100 000 pregnant women. If our estimates of the number of pregnancies are reasonably accurate, then 1 listeriosis infection occurs in every 22 000 pregnancies in the population. Among Hispanics, we estimate 1 listeriosis infection in every 8000 pregnancies. Almost one-third of cases resulted in fetal loss or neonatal death.
Effective prevention among older adults also would have a substantial impact on overall rates, given that rates in this age group are 4–5 times higher than the general population. Listeriosis among adults aged ≥65 years accounted for more than half of all listeriosis cases and caused 79 deaths. These results are significant because older adults represent a large and growing demographic group in the United States, approximately 40 million persons [24]. Including persons aged 45–64 years and those with certain medical conditions [19], the population of persons at higher risk for nonpregnancy-associated listeriosis is even larger. In fact, the analyses by Pouillot et al suggest that a single age cutoff to define older adults (eg, 65 years) may be overly simplistic for risk assessments and related purposes requiring nuanced descriptions of variations in risk across population groups [19]. Another important limitation of these data is that the specific risks of nonpregnancy-associated listeriosis attributed to older age versus an immunocompromised or debilitated underlying health status could not be distinguished because of incomplete data on comorbid conditions.
Overall, the incidence of listeriosis did not change significantly from 2004 through 2009. This finding contrasts with significant declines from 1989 through 2000, which were described previously using data from similar population-based surveillance systems [1, 13]. Industry, regulatory, and educational efforts to prevent listeriosis in the United States contributed to declines during the 1990s [1, 13]. After 1989, when a relatively high overall incidence of 0.79 cases per 100 000 was documented by the surveillance system preceding FoodNet, population-based incidence estimates declined by two-thirds to a mean of 0.28 cases per 100 000 from 2000 through 2009. In contrast, estimates of the overall incidence of listeriosis in 2000 and 2009 are similar (0.33 and 0.32 cases per 100 000, respectively), suggesting that those efforts may have reached the limit of their impact [1, 13]. The lack of recent progress indicates that new industry and regulatory strategies and a renewed commitment to prevention will be necessary to reduce the incidence of listeriosis further. Based on our data, we conclude that groups with the highest incidence, pregnant women (especially Hispanics) and older adults, should be prioritized for prevention efforts, including targeted improvements in the safety of higher-risk foods and continuing, food safety education [25].
Although we could not identify the specific food items that caused these infections, patients in higher-risk groups frequently reported consumption of foods previously implicated in listeriosis outbreaks and L. monocytogenes risk assessments [26]. For example, Hispanic patients frequently consumed Mexican-style soft cheese (queso fresco), which has been implicated in numerous listeriosis outbreaks caused by the use of unpasteurized milk for manufacturing cheese [11, 12]. Outbreaks involving postprocessing contamination of Mexican-style cheese made from pasteurized milk also have occurred recently in the United States [27], but whether pasteurized milk was used for the cheeses consumed by Hispanic patients in our report is unknown. Ongoing research, policy, and prevention efforts are needed to address the safety of this popular food during and after manufacturing [28]. Recent outbreaks of hospital-acquired listeriosis also indicate that combined interventions are needed to improve food safety in hospitals and healthcare settings where older adults and other vulnerable patients receive care [29]. Studies in Australia and the United Kingdom have demonstrated that patients with listeriosis had frequently (40%) been exposed to foods known to be at risk for L. monocytogenes contamination during a prior hospitalization and that sandwiches served in healthcare settings had a substantial prevalence (3%) of L. monocytogenes contamination [30, 31].
Numerous resources, such as food safety guidelines, educational materials, and messaging strategies, as well as a better understanding of related knowledge, attitudes, and behaviors have already been developed specifically for many of the populations at risk for listeriosis, including pregnant women [32], persons with HIV/AIDS [33], transplant recipients [34], patients with cancer [35], and older adults [36]. State and local health departments as well as other stakeholders (eg, other government agencies, community-based organizations, academic institutions, and healthcare and care-giving organizations) should work collaboratively and creatively to use these materials and knowledge to educate pregnant women, Hispanics, persons with immunocompromising or chronic medical conditions, and older adults on prevention of listeriosis. Dieticians, geriatricians, infectious disease specialists, nephrologists, nurse practitioners, obstetricians, oncologists, and other healthcare providers are uniquely positioned to provide credible information to an engaged and available patient audience that may be motivated to learn during a period of heightened risk. Although clinicians probably already appreciate the severity of invasive listeriosis, an emphasis on their important role as educators might substantially enhance the delivery of food safety education [37]. Internet-based resources developed by multiple US federal health agencies and currently available online have been consolidated in the Supplementary Data.
The seasonality that we observed in nonpregnancy-associated listeriosis is consistent with 2 other reports of listeriosis-related morbidity and mortality in the United States [1, 38]. Public health officials, clinicians, and other health educators should consider intensifying food safety education campaigns during the summer months. The reason seasonality is seen only in non–pregnancy-associated listeriosis is unknown, but the phenomenon may offer important clues about foodborne transmission of listeriosis, such as temporal variation in environmental sources of contamination and the risks of foods that are consumed seasonally.
We have probably underestimated the true incidence of listeriosis because about half of the cases that actually occur may not be diagnosed [2]. In addition, invasive listeriosis is a rare disease, which makes precise estimation of incidence difficult, especially for relatively small subpopulations like pregnant Hispanic women and older adults. The nonrandom selection of sites that participate in FoodNet [16] and the fact that the incidence of pregnancy- and nonpregnancy-associated listeriosis varied 20- and 4-fold, respectively, among surveillance sites suggest that national extrapolation is contingent on strong assumptions of representativeness. Furthermore, states with an incidence of pregnancy-associated listeriosis that was higher than the median did not necessarily have a relatively higher incidence of nonpregnancy-associated listeriosis (data not shown). Adjusted incidence estimates accounted for this variability appropriately but further diminished the precision of specific incidence estimates. These statistical limitations made it particularly difficult to assess the significance of the 2007–2009 increase in pregnancy-associated listeriosis. Although the increase was not significant when adjusted incidence rates were modeled for Hispanic and non-Hispanic pregnant women, increases were consistently observed in each of the 3 years and occurred in all but the 2 states that had the lowest rates of pregnancy-associated listeriosis (New Mexico and Minnesota). Our crude incidence rate estimates describe the absolute risk of listeriosis in key population subgroups over time, essential information for clinicians and public health practitioners. However, these estimates cannot fully elucidate the interactions between demographic variables; the results of relative risk modeling conducted by Pouillot et al show that important interactions do exist (eg, lower risk for Hispanics aged 60–69 years compared with non-Hispanics in the same age group) [19].
Efforts to reduce the incidence of listeriosis were successful during the 1990s, but progress has slowed in the last decade. The Healthy People 2005 and 2010 national objective of ≤0.24 cases per 100 000 population [14] was not met, and new strategies are needed to achieve the Healthy People 2020 objective [39], which strives for further overall reductions (≤0.20 cases per 100 000). As the national population of older adults and persons living with immunosuppression continues to grow, public health interventions that target the foods consumed by these and other groups at higher risk for listeriosis, including pregnant women, will be critical to protect these populations and decrease the overall incidence of listeriosis.
Notes
Acknowledgments.
Effie Booth, Shaun Cosgrove, Stacy Crim, Ryan Gierke, Sharon Greene, Theodore Harper, Julie Hatch, Robert M. Hoekstra, Karunya Manikonda, Mickey Parish, Mary E. Patrick, Charlie Saltalamacchia, Suzanne Segler, Suzanne Solghan, Melissa Tobin-D’Angelo, and Madhavi Vajani provided technical assistance with the surveillance data sets, critical review of the manuscript, or both. We thank our colleagues in state and local health epidemiology and laboratory programs and the numerous clinicians and microbiologists whose collaboration makes FoodNet surveillance possible.
Disclaimer.
The contents of this work are solely the responsibility of the authors and do not necessarily represent the official views of the Centers for Disease Control and Prevention.
Financial support.
This publication was supported in part by the Centers for Disease Control and Prevention (CDC; Cooperative Agreement U60/CD303019). FoodNet is funded by the Food Safety Office and the Emerging Infections Program of CDC, the US Department of Agriculture Food Safety and Inspection Service, and the Food and Drug Administration.
Supplement sponsorship.
This article was published as part of a supplement entitled “Studies From the Foodborne Diseases Active Surveillance Network,” sponsored by the Division of Foodborne, Waterborne, and Environmental Diseases of the National Center for Emerging and Zoonotic Infectious Diseases from the Centers for Disease Control and Prevention, and the Association of Public Health Laboratories.
Potential conflicts of interest.
All authors: No reported conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.





Comments